This site is intended for US healthcare professionals only.
I am a US healthcare professional and would like to continue.
Healthcare professionals outside of the US: click here.
Sign Up
Join the list to be notified about Rayaldee news
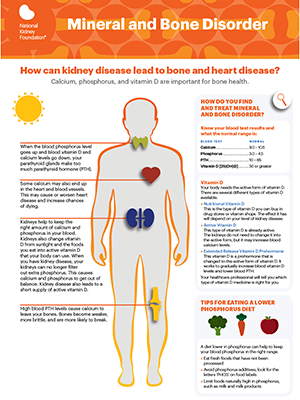
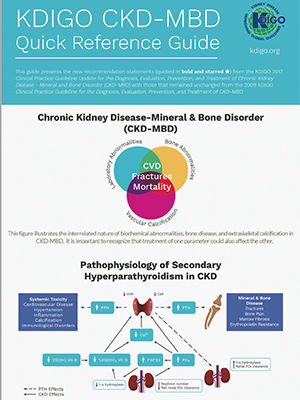
Declaration of consent OPKO Pharmaceuticals LLC wishes to use your email address and/or phone number to periodically send you emails and/or text messages containing information and company news related to chronic kidney disease (CKD) and secondary hyperparathyroidism (SHPT). Message frequency may vary. You may opt out at any time. Standard message and data rates may apply.
Terms of Use and Privacy Policy
Healthcare professionals outside of the US: click here.